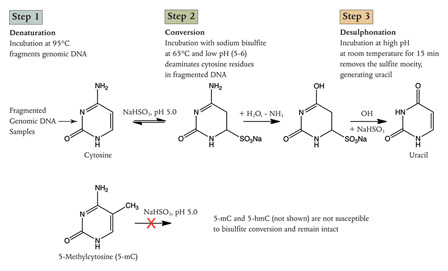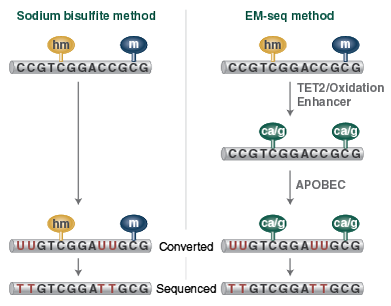
Bisulfite Conversion
Choose Type:
- What types of sample can be processed using the NEBNext® Enzymatic Methyl-seq Kit and the Enzymatic Methyl-seq Conversion Module?
- How should EM-seq sequencing data be analyzed?
- How should I store my bisulfite converted DNA after it is eluted from the column?
- No PCR product is detected when I amplify more than 300 bp amplicon.
- What is the shelf life of the reagents supplied?
- Which DNA polymerase do you recommend for amplification of converted DNA?
- Does my sample need to be free of RNA for bisulfite conversion?
- Do I need extra purification of my mammalian DNA prep in order to use it in a bisulfite conversion reaction?
- My thermocycler does not allow to set up the reaction volume to 140 μl, what I should do?
- Capture and Elute Step for Control DNA (E2600)
- Capture Methylated CpG DNA (E2600)
- Cycling Protocol Using EpiMark™ Bisulfite Conversion Kit (E3318)
- DNA Fragmentation (E2600)
- Downstream Analysis (NEB #E2600)
- Elute Captured Methylated CpG DNA (E2600)
- End-point PCR Using EpiMark Bisulfite Conversion Kit (E3318)
- Optimizing Restriction Endonuclease Reactions
- Prebind MBD2a-Fc to Protein A Magnetic Beads (NEB #E2600)
- Protocol for use with NEBNext ChIP-seq Library Prep Reagent Set for Illumina (E6200)
- Reagent Preparation Using EpiMark® Bisulfite Conversion Kit (E3318)
- Wash Off Unbound DNA (E2600)
-
Epigenetics - Expanding on Genomic Foundations
- Epigenetics Brochure
Feature Articles
Brochures
- Chernov AV., Reyes L., Peterson S., Strongin AY. (2015) Depletion of CG-Specific Methylation in Mycoplasma hyorhinis Genomic DNA after Host Cell Invasion PLoS One; 10, e0142529. PubMedID: 26544880, DOI: 10.1371/journal.pone.0142529
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.
This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.